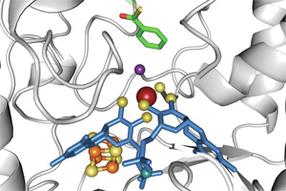
 Scientists of the Freiburg University have discovered how bacteria break down aromatic compounds in anaerobic conditions. The bacteria use an enzyme which contains a tungsten atom (dark red) to destabilize an aromatic ring (green).
Tungsten is the heaviest metal with a biological function.
Because aromatic compounds are very common in nature, it is important to know how they are recycled in the carbon cycle. This is especially the case since crude oil contains such compounds which are hard to break down and often toxic and carcinogenic.
Scientists of the Freiburg University have discovered how bacteria break down aromatic compounds in anaerobic conditions. The bacteria use an enzyme which contains a tungsten atom (dark red) to destabilize an aromatic ring (green).
Tungsten is the heaviest metal with a biological function.
Because aromatic compounds are very common in nature, it is important to know how they are recycled in the carbon cycle. This is especially the case since crude oil contains such compounds which are hard to break down and often toxic and carcinogenic.
How bacteria break down aromatic compounds in environments without oxygen was however unknown until now. Such environments can be river and lake sediments, contaminated ground water or biogas installations. Knowing tungsten's key role may enable addition of tungsten(compounds) to help bacteria break down aromatic contamination, or increase energy production in biogas installations. Also tungsten containing biocatalysts may be developed that can be used to produce specific compounds out of aromatic compounds that currently can only be produced using toxic substances.
 Wetenschappers van de Universiteit Freiburg hebben het enzym ontdekt waarmee bacteriën benzeenringen zonder zuurstof afbreken. Benzeenringen, die kenmerkend zijn voor zogenaamde aromatische verbindingen, zijn zeer stabiele verbindingen die moeilijk biologisch afbreekbaar zijn.
Wetenschappers van de Universiteit Freiburg hebben het enzym ontdekt waarmee bacteriën benzeenringen zonder zuurstof afbreken. Benzeenringen, die kenmerkend zijn voor zogenaamde aromatische verbindingen, zijn zeer stabiele verbindingen die moeilijk biologisch afbreekbaar zijn.
De Freiburgse onderzoeksgroep laat in een artikel in "Nature Chemical Biology" zien hoe in een reactie met een enzym met een wolfraamatoom (donkerrood) in het actieve centrum een aromatische ring wordt gedestabiliseerd.
Wolfraam is het zwaarste metaal met een biologische functie. Omdat benzeenringen in de natuur veel voorkomen, is het van groot belang hoe deze structuren in de wereldwijde koolstofcyclus worden gerecycled. Dat geldt met name omdat aardolie veel benzeenverbindingen bevat, die vaak giftig en kankerverwekkend zijn.
Aromatische benzeenringen worden vooral in houtplanten gevormd en verspreiden een karakteristiek aroma. Al lange tijd is bekend dat zuurstofminnende bacteriën deze ringen met behulp van zuurstof afbreken. Hoe bacteriën dit in een omgeving zonder zuurstof doen was echter niet bekend. Denk daarbij aan sedimenten van meren en rivieren, vervuild grondwater, of biogasinstallaties.
Nu we weten welke cruciale rol wolfraam speelt, biedt dit interessante toepassingsmogelijkheden. Toevoeging van wolfraam of wolfraamverbindingen kan mogelijk helpen bij de afbraak van bodemverontreiniging, of kan mogelijk de opbrengst van biogasinstallaties verhogen. Mogelijk kunnen ook biokatalysatoren worden ontwikkeld die in biotechnologische processen aromaten omzetten in specifieke verbindingen, die anders alleen met toepassing van zeer giftige substanties te maken zijn.
 Scientists of the Freiburg University have discovered how bacteria break down aromatic compounds in anaerobic conditions. The bacteria use an enzyme which contains a tungsten atom (dark red) to destabilize an aromatic ring (green).
Scientists of the Freiburg University have discovered how bacteria break down aromatic compounds in anaerobic conditions. The bacteria use an enzyme which contains a tungsten atom (dark red) to destabilize an aromatic ring (green).